In the previous article, I chatted with Dr. Sierra on the principles of Proximity Ligation Assay (PLA).
Continuing with our conversation, Dr. Sierra went into the technical aspects of running a PLA and provided some bench tips and ways to troubleshoot.
General Tips
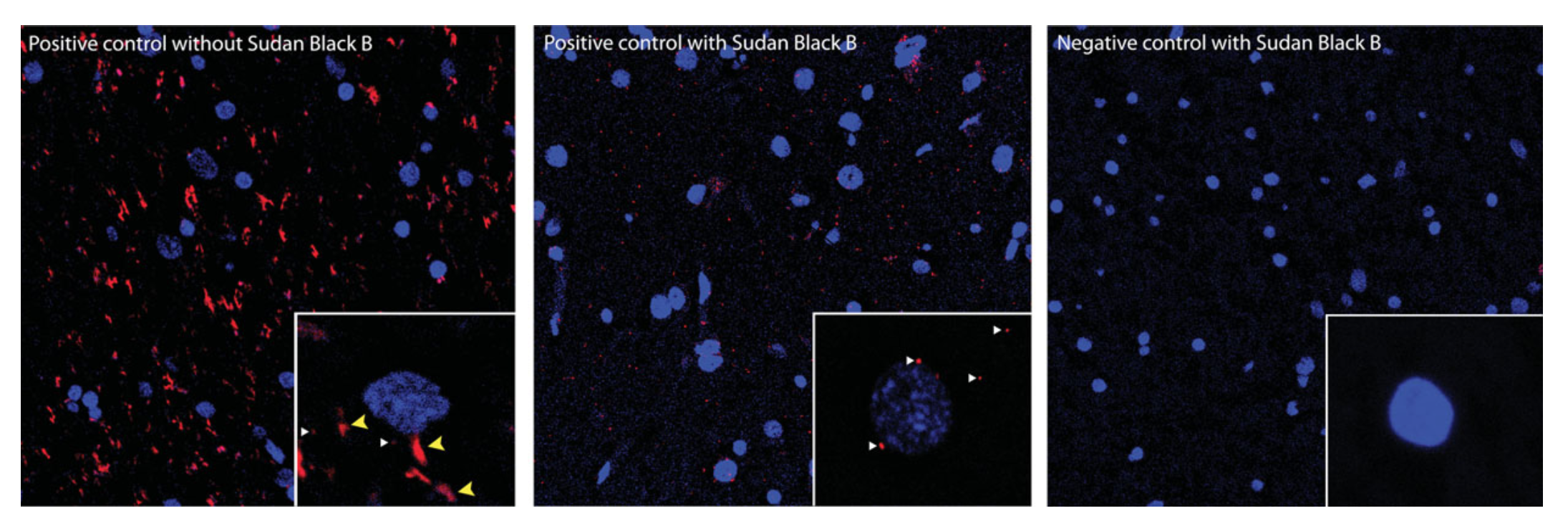
- When fixing cells with paraformaldehyde (PFA), incubate the sample in glysine to quench the aldehyde groups to reduce autofluorescence that could interfere with the PLA signal. When using PFA or formalin-fixed mice and human brain samples, use Sudan Black B to quench PFA or lipofuscin-like autofluorescence.
- Make sure to include both positive and negative controls to validate the PLA signal observed in your sample.
- To ensure validity of the PLA data, it's recommended to use 3 tissue slices from 3 individual animals per treatment. For cells, triplicate wells should be used for each treatment, with the experiment repeated 3 times.
- Antibodies that work well with IF will usually work well in PLA.
- Avoid using PLA probe-conjugated secondary antibodies that target the IgG of the same species as your sample. (ie. Don't use anti-mouse PLA probes in mouse tissue.)
- It's best to use red fluorophore (568nm) for the PLA signal as it is more stable and easier to visualize.
- Slides with PLA signal can be stored in the dark for 15 days at 4 degrees Celsius, and for months at -20 degrees. However, as the PLA signal is light sensitive it is best to take the measurement as soon as possible.
Image Source: Figure 2.16.5, Gomes et al. (2016) Curr. Protoc. Pharmacol.
Tips by Step
Here are some steps to watch out for in the PLA protocol:
The Slides
- Mouse tissue: Gelatin makes it easier to mount the slides. Consider using 0.2% gelatin in 0.05M Tris-Cl, pH 7.6 buffer instead of PBS.
- Cells: the Lab-Tek chamber slides is easy work with and prevents cells from drying up, which can affect PLA signal.
- FFPE Human Brain Sample: Make sure to perform antigen retrieval prior to treating the slide with PLA reagents.
- Use an hyprophobic pen to encircle your sample as it helps to keep the reagents on your sample.
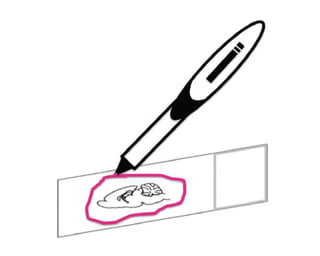 Like so
Like so
Blocking
Blocking solution (50-70uL) should be added when the slides are placed in the humidity chamber and the incubator.
When removing blocking solution, make sure the sample is never completely dry.
Applying Primary Antibodies
Only use antibodies from a mouse, rabbit, or goat source, as commercially available PLA probes only target the IgG of these species.
Applying PLA Probes
It does not matter which secondary antibody is conjugated to the PLUS or MINUS, as long as both PLUS and MINUS are used.
Signal Amplification
Make certain that the slides are kept in the dark as the fluorophore added during amplification is light sensitive.
Image Acquisition and Signal Quantification
Confocal microscopes produces higher resolution images than epifluorescence microscopes. It allows for imaging a thin slice out of a thick specimen, enhancing the signal count through the entire cell. The z-stacking of confocal microscopy also facilitates the visualization of individual PLA signals.
While many software applications are commercially available for PLA measurement and quantification, they are often quite expensive. ImageJ with the Iterative Deconvolve 3-D plugin is a cheaper alternative.
Images should be captured at 40x magnification. It is best to image at least 30 cells per experiment, each cell cropped and saved individually, so parametric tests can be used for statistical analysis.
Troubleshooting and Solutions
| Problem | Causes | Solutions |
| No PLA Signal | Tissue slice/cells dried out during assay | Use a hydrophobic pen to draw a line around the sample to keep reagent in place |
| Protein levels to be decteced might be low | Increase 1' antibody concentration | |
| High Background | High concentration of 1' antibodies | Decrease 1' antibody concentration |
| Amplification carried out for too long | Do not exceed 100min for amplification | |
| Microscope settings need to be adjusted | Do not overexpose samepl to light; use conocal microscope | |
| Presence of dust, fixative, or salt precipitation | Use new reagents and add additional washes with new wash solutions; change blocking solution; make certain there are no autofluorescent particles such as lipofuscin | |
| Wash buffers improperly prepared | Follow recipe as described; do not use Tris base instead of Tri hydrochloride | |
| Poor animal perfusion | Use tissue from another animal | |
| Tissue slice too thick | Use thin slices (40uM or less) or use confocal microscopy to adjust Z-stack | |
| 1' antibodies conjugated with PLA riboprobes fail to work | Design experiments to use the entire amount of 1' antibodies lined to PLA riboprobe or store them at 4 degree Celsius for less than 1 week. |
Data source: Table 2.16.1, Gomes et al. (2016) Curr. Protoc. Pharmacol.
For detailed protocol for Direct and Indirect PLA using cells, animal tissues, and FFPE human brain samples, as well as buffer recipes, please refer to Dr. Sierra's paper in 2016. You can also watch his recent talk on PLA here:
I hope you have found this series on PLA useful.
If you're planning a PLA experiment, try out BenchSci and examine published PLA data to help you find the most suitable antibodies for your study.